Gallinazo
A towering pioneer whose lavender crown bursts above the canopy like a signal flare. The Galibi people of French Guiana gave it the name "copaia," and two centuries of European botanists tried to replace it with Latin.

When Jacaranda copaia flowers, it does so with an extravagance matched by few other canopy trees. The entire crown erupts in blue-lavender blooms, a mass of color so vivid it stands out above the forest canopy for kilometers. In Costa Rica, where it is known as gallinazo, the display occurs between February and April. In the central Amazon, it comes later, from August through November, during the dry season when the tree directs energy stored from the previous year's rains into flower production. A single tree may bloom for four weeks, but only about one in every hundred flowers sets fruit. The tree is an obligate outcrosser, dependent on Euglossa and Centris bees that arrive at dawn to work the tubular corollas.
The name copaia comes from the Galibi (Carib) people of French Guiana, who used the tree medicinally and knew it long before any European pharmacist catalogued it. French colonists called it "onguent pian" because they applied preparations of its bark and leaves against yaws, a tropical skin disease. When the pharmacist-botanist Jean-Baptiste Aublet described it in 1775, he kept the Galibi name. Later European botanists, uncomfortable with what they considered a "barbaric" epithet, tried to replace it with the Latin procera ("tall"). The replacement failed. Aublet's indigenous name stands as the accepted epithet to this day, a small victory for the knowledge of the people who understood the tree first.
Identification
Habit

Jacaranda copaia is a large canopy to emergent tree, reaching 30 to 45 m tall with a cylindrical trunk up to 50-90 cm in diameter, unbranched for more than half its height. It is the only member of the genus Jacaranda that grows to full canopy stature in rainforest, and it has the largest wood vessels of any tree in the Bignoniaceae. Young trees are immediately recognizable by a distinctive growth form: a slender, unbranched trunk topped by a terminal plume of ascending branches bearing enormous bipinnate leaves, an architecture that resembles a tree fern. This youthful form is so striking that it can be confused with the unrelated legume Schizolobium parahyba, which adopts a near-identical habit in the same lowland secondary forests. Mature trees develop a vase-shaped or plume-like crown. The trunk is basally swollen but lacks buttresses.
Leaves

The leaves are opposite and bipinnate (doubly compound), among the largest in the Bignoniaceae at 30 to 160 cm long and up to 60 cm wide. The petiole measures 4-29 cm, and the rachis (central axis) is narrowly grooved above but lacks the lateral wings found in the related J. caucana. Each leaf bears 5 to 20 pairs of pinnae, each 7-35 cm long. The leaflets are 20-80 mm long and 6-18 mm wide, asymmetrically rhomboid-elliptic to elliptic-oblong, with an acuminate (tapering) apex and a lopsided base: one side narrowly cuneate (wedge-shaped), the other broadly obtuse. Secondary veins number 3-7 per side. The upper surface is dark when dried, with minute hairs along the midvein; the lower surface is sparsely puberulent (finely hairy) on the veins. The texture is chartaceous (papery).
Flowers

The inflorescences are open pyramidal panicles up to 48 cm long, with lateral branches reaching 30 cm. Each bears many flowers on short pedicels (stalks). The calyx is tubular-cupular (cup-shaped), 4-7 mm long and 3-5 mm in diameter, densely covered in minute hairs and irregularly lobed or nearly entire at the margin. This cupular calyx is unique within the genus and one of the diagnostic features that places J. copaia in its own section, Jacaranda sect. Copaia. The corolla is tubular-campanulate (bell-shaped above a narrow base), 23-50 mm long, blue or lavender with a white throat, and densely puberulent on the exterior. Four stamens bear monothecal (single-chambered) anthers, another feature distinguishing this section. The staminode (sterile stamen) is 24-27 mm long, pubescent, and bifurcate (forked) at the tip. In Costa Rica, flowering occurs from February to April; in the central Amazon, from August to November during the dry season.
Fruits
The fruits are woody capsules, 4.5 to 12 cm long and 3 to 6 cm wide, oblong-rounded with a smooth but minutely muricate (rough-textured) surface. They are flat, only 5-8 mm thick, and split open perpendicular to the seed-bearing axis (loculicidal dehiscence). Each capsule can contain up to 250 seeds. The seeds are 11-14 mm long and 21-30 mm wide, with a small central body about 5 mm in diameter surrounded by translucent membranous wings bearing distinctive brown rays. These winged seeds disperse by wind. A genetic study on Barro Colorado Island, Panama, tracked seed movement using microsatellite markers and found that about 76% of seeds landed within 100 m of the mother tree, while 20% traveled farther, with a maximum recorded distance of 710.5 m. Seeds germinate within a few days of landing. In Costa Rica, fruiting occurs from July to November.
Trunk and Bark

The bark is light brown and corky when young, becoming gray and smooth with age, and bearing pale lenticels (pores). The twigs are subtetragonal (nearly four-angled), lepidote (scaly), and dry to brown or blackish. They contain a large pith that is sometimes hollowed out by ants. The trunk base is swollen without true buttresses. The wood itself is remarkably light, with a basic specific gravity of only 0.35 and an air-dry density of 0.3-0.52 g/cm3. The heartwood and sapwood are poorly differentiated, ranging from dull white to oatmeal with prominent brown vessel lines. Despite its softness, the wood has a high luster and straight grain, and it peels and slices into veneer easily. It is used for decorative boards, particleboard, furniture, pulp, pencils, matches, and musical instruments, and the International Tropical Timber Organization reports that it is exported regularly from Brazil to China and Japan.
Herbarium Specimens

Distribution

Jacaranda copaia ranges from southern Mexico through Central America to Bolivia and Brazil, spanning at least ten countries. Colombia holds the majority of GBIF occurrence records (nearly 60%), followed by Brazil (16%), French Guiana, Peru, Venezuela, Panama, Guyana, Ecuador, Suriname, and Bolivia. The species grows in lowland wet tropical forests from near sea level to about 850 m, preferring well-drained upland (terra firme) sites and avoiding seasonally flooded areas. A landmark 2013 study in Science classified it among the 227 "hyperdominant" species that account for half of all individual trees in Amazonia, with local densities exceeding 60 trees per hectare in some plots.
In Costa Rica, J. copaia occurs on both the Caribbean and Pacific slopes, from 10 to 600 m elevation, in humid and very humid forest types. GBIF records 65 occurrences across 36 localities spanning all seven provinces. The Caribbean slope records include La Selva Biological Station in Heredia, Estacion Pitilla in Guanacaste (at 850 m, the upper limit of its Costa Rican range), and Bosque Lluvioso near Guapiles in Limon. On the Pacific side, collections come from the Peninsula de Osa, the Valle de General near San Isidro, and Parque Nacional La Cangreja in San Jose Province. In the Brunca (South Pacific) region, seven localities are documented, concentrated along the Golfo Dulce coast: the Reserva Forestal Golfo Dulce near Rincon, Bajo de San Juan along the Rio Chocuaco, La Gamba near kilometer 37, and the Osa Peninsula near the Rincon airstrip, where collections date back to 1971. The species is documented at the Osa Arboretum.
Ecology
Jacaranda copaia is a classic long-lived pioneer. It colonizes canopy gaps and recently cleared areas, depending on high light for recruitment, yet persists in mature forest as a canopy or emergent tree. Studies in French Guiana found clumps of genetically related saplings in canopy gaps, indicating that clusters of siblings establish together when a gap opens, but no seedlings survive under closed canopy. This strategy makes it one of the fastest-growing trees in Neotropical rain forests. Finegan et al. (1999) classified it among the fast or very fast growers in a study of 106 tree species in a logged Costa Rican rain forest. Trees reach reproductive maturity in about 10-15 years, and in French Guiana, produce annual growth rings visible to the naked eye. Growth is positively correlated with wet-season precipitation.
The species is an obligate outcrosser with late-acting self-incompatibility. In a pollination study at the Floresta Nacional do Tapajos in the central Amazon, Maues et al. (2008) documented about 40 species of native bees visiting the flowers, but identified medium-sized solitary bees in the genera Euglossa (orchid bees) and Centris as the primary pollinators. Their body size matches the corolla tube diameter, ensuring direct contact with the reproductive structures. Flowers open at dawn, when these bees are most active. Despite heavy visitation, natural fruit set is remarkably low: only 1.06% of open-pollinated flowers produced fruit (from 6,932 flowers monitored). Hand cross-pollination raised this to 6.54%, while self-pollination produced zero fruits. This "cornucopia" flowering strategy, flooding pollinators with more flowers than they can visit, ensures outcrossing at the cost of massive flower overproduction.
Seeds are wind-dispersed, released during the season of maximal canopy gap creation, when sunlight and light winds coincide. Jones et al. (2005) used genetic parentage analysis to track dispersal in a fully mapped 50-hectare plot on Barro Colorado Island, Panama. Most seeds (76%) fell within 100 m of their mother, consistent with a gravity-assisted wind dispersal pattern, but a substantial tail of long-distance events carried seeds up to 710.5 m. These rare long-distance events are ecologically significant: they connect isolated gap populations and maintain genetic diversity across the landscape. The species' rapid germination, often within days of seed release, allows it to exploit freshly opened gaps before competitors.
Taxonomic History
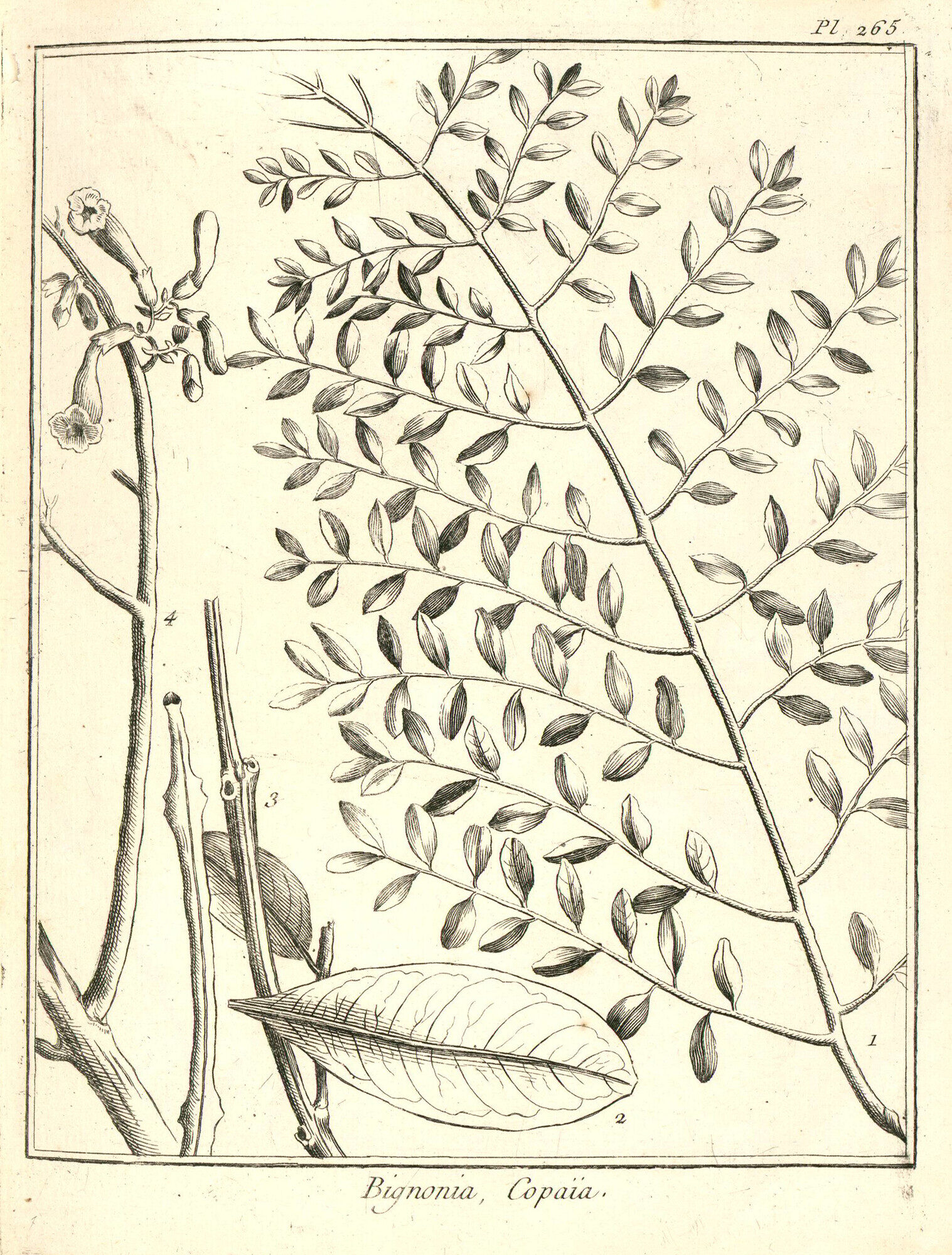
Jean-Baptiste Christophore Fusee Aublet (1720-1778) first described this species as Bignonia copaia in his landmark Histoire des Plantes de la Guiane Francoise (1775, volume 2, page 650), illustrated on plates 262 and 265. Aublet was a French pharmacist who traveled to French Guiana as part of the Kourou Expedition of 1762-1764 to investigate the colony's botanical resources. He worked alongside Amerindian guides, enslaved Africans, and European colonists, and the resulting work described more than 400 species new to science, with many epithets drawn from indigenous Galibi and Creole names. When Aublet died in Paris in 1778, he bequeathed part of his plant collection to Jean-Jacques Rousseau, who held it for only two months before also dying. The herbarium eventually reached the Museum national d'Histoire naturelle in 1953.
In 1823, the Scottish botanist David Don (1799-1841) transferred the species to the genus Jacaranda in the Edinburgh Philosophical Journal (volume 9, page 267), creating the currently accepted combination Jacaranda copaia (Aubl.) D. Don. Don was librarian to the Linnean Society from 1822 and later professor of botany at King's College, London. The genus name Jacaranda comes from the Tupi-Guarani languages of South America, likely meaning "fragrant" or derived from y-aca-rata ("heartwood"). Several synonyms accumulated over the centuries: Carl Ludwig Willdenow published Bignonia procera in 1800, and Robert Brown independently published Jacaranda procera in 1822, both superfluous names intended to replace Aublet's Galibi-derived epithet with something more conventionally Latin. As Zarucchi (1984) noted, European botanists of the era considered indigenous-derived names "barbaric" and routinely replaced them. Other synonyms include Jacaranda spectabilis Mart. ex DC. (1845), which became the basis for subspecies spectabilis when Alwyn Gentry formally recognized it in 1977, and Jacaranda superba Pittier (1940).
A 2019 phylogenetic study by Fonseca and Lohmann placed J. copaia in its own section within the genus: Jacaranda sect. Copaia, defined by the unique combination of monothecal anthers (single-chambered, unlike the dithecal anthers of sect. Dilobos) and unlobed cupular calyces (unlike the campanulate calyces of sect. Jacaranda). The species also has distinctive wood anatomy, with multiseriate homocellular rays intermediate between the other two sections and the largest wood vessels in the entire family Bignoniaceae. Two subspecies are currently recognized: subsp. copaia, with smaller leaves bearing 5-9 pinnae and symmetric leaflet bases, and subsp. spectabilis, with larger leaves, up to 20 pinnae, and asymmetric leaflet bases. Molecular data from Scotti-Saintagne et al. (2013) supports this subdivision, showing that the two subspecies correspond to distinct genetic clusters, though nuclear admixture between them confirms they belong to a single species rather than two separate ones.
Biogeography
The phylogeographic history of J. copaia is one of the best-studied of any Neotropical tree. Scotti-Saintagne et al. (2013) analyzed 341 adult trees across the species' range using nuclear microsatellites, chloroplast microsatellites, and cpDNA sequences. They found that chloroplast DNA haplotype diversification began at the end of the Pliocene, about 2.61 million years ago, with the central Amazon Basin harboring the highest haplotype diversity, the likely center of origin. Population divergence was more recent, occurring during the mid-Quaternary (357,000-436,000 years ago). A key finding was that chloroplast diversity did not show the expected east-west split across the Andes, suggesting that the species crossed the Andean barrier through recent dispersal rather than being separated by the mountains' uplift. This cross-Andean dispersal may have been facilitated by the species' wind-dispersed seeds and its pioneer ecology, which allows rapid colonization of newly available habitat.
Similar Species

Three Jacaranda species occur in Costa Rica. All three produce blue-to-lavender flowers during the dry season, but they differ in how they display them. J. copaia flowers among its leaves, its large panicles (to 48 cm) opening within the standing foliage from February to April. Jacaranda caucana Pittier, the other native species, sheds its leaves before flowering (March to May), producing compact panicles (6-15 cm) of purple-blue flowers on bare branch tips. Jacaranda mimosifolia D. Don, the widely planted ornamental native to Argentina and Bolivia, also flowers on leafless or nearly leafless branches (January to July, peaking April-May), creating the familiar lavender canopy seen in parks and streetscapes. In flower structure, the three species are distinguished by the calyx: J. copaia has a deep tubular-cupular calyx (4-7 mm), unique in the genus; J. caucana has a short, widely flaring campanulate calyx (1.5-3 mm); and J. mimosifolia has a tiny toothed cup (1-2 mm). The corolla of J. caucana is often bent almost 90 degrees near the base, and the Costa Rican subspecies (sandwithiana) has glandular-pilose hairs on the corolla exterior, a different pubescence from the dense minute puberulence of J. copaia. Beyond flowers, J. caucana is a smaller tree (8-28 m) with smaller leaflets (8-28 mm vs. 20-80 mm in J. copaia) and fruit capsules with undulating margins (vs. straight). J. mimosifolia is smaller still (8-15 m) with tiny leaflets (5-14 mm long) and hard, rounded, lens-shaped fruits unlike the flat oblong capsules of J. copaia. The unrelated legume Schizolobium parahyba closely mimics the growth habit of young J. copaia, both producing slender unbranched trunks with terminal tufts of large compound leaves in lowland secondary growth. They can be distinguished by leaf arrangement: opposite in Jacaranda, alternate in Schizolobium, and by leaflet shape: asymmetric in Jacaranda, rounded in Schizolobium.
Chemistry and Traditional Uses
The Galibi name "copaia" and the French colonial name "onguent pian" (yaws ointment) both point to the tree's long medicinal history. Across its range, indigenous and rural communities have applied bark and leaf preparations to treat skin infections, wounds, bronchitis, fevers, and rheumatism. The Andoque people of the Colombian Amazon prepare a decoction of crushed leaves reduced to a honey-like consistency for topical wound healing. Leaves and wood are also burned to produce smoke that repels mosquitoes. The flat seed pods serve as tools for shaping pottery.
Modern chemistry has validated some of these traditional uses. Sauvain et al. (1993) isolated ursolic acid, a pentacyclic triterpene, from leaf extracts and demonstrated activity against Leishmania amazonensis at an ED50 of 20 micromolar without toxicity to macrophages, relevant to the tree's traditional use against skin diseases including leishmaniasis. The compound jacaranone, a cytotoxic semibenzoquinone first isolated from the related J. caucana by Ogura, Cordell, and Farnsworth in 1976, was patented for antitumor activity in 1977 after showing activity against P-388 lymphocytic leukemia in mice at 2 mg/kg. More recent studies have tested jacaranone derivatives against breast, cervical, lung, liver, and colon cancer cell lines, with antiproliferative IC50 values ranging from 6.3 to 26.5 micromolar. An ethnopharmacological review by Gachet and Schuhly (2009) catalogued antioxidant, antimicrobial, anticancer, anti-leishmanial, and hypotensive activities across the genus.
Conservation Outlook
Jacaranda copaia is assessed as Least Concern on the IUCN Red List (2020, taxon ID 61985587). The assessment reflects its wide distribution across at least ten countries, its large and stable population, its classification as a hyperdominant Amazonian species, and its vigorous pioneer ecology, which allows it to regenerate rapidly in disturbed habitats. The species is documented from numerous protected areas including the Floresta Nacional do Tapajos (Brazil), Barro Colorado Island (Panama), and Area de Conservacion Guanacaste (Costa Rica). No major threats currently affect the species, though ongoing deforestation and agricultural expansion in Amazonia and Central America reduce available habitat. Its ability to colonize clearings and secondary growth mitigates this pressure to some extent, making it one of the more resilient canopy trees in the Neotropics.
Resources & Further Reading
Species Information
Plants of the World Online entry with distribution, synonymy, and IUCN status.
Global occurrence records (7,882 records across 10 countries) and specimen data.
General information including taxonomy, description, and distribution.
Detailed species account including traditional uses, wood properties, and cultivation.
Technical data on wood properties, density, hardness, and workability.
Taxonomy & Nomenclature
Nomenclatural data, 46 synonyms, and specimen records from Missouri Botanical Garden.
Molecular phylogenetic study establishing sect. Copaia. American Journal of Botany.
Range-wide phylogeographic analysis using 341 trees. Journal of Biogeography 40: 707-719.
Ecology & Conservation
Pollination study at Floresta Nacional do Tapajos, documenting Euglossa and Centris bees. Revista Brasileira de Botanica.
Genetic parentage analysis of seed dispersal on Barro Colorado Island, Panama. The American Naturalist 166(5): 543-555.
Landmark study classifying J. copaia as a hyperdominant Amazonian species. Science.
Diameter increment study of 106 tree species including J. copaia. Forest Ecology and Management 121(3): 159-176.
Chemistry & Ethnobotany
Comprehensive review of traditional uses and phytochemistry across the genus. Journal of Ethnopharmacology.
First isolation of the anticancer compound jacaranone. Lloydia 39: 255-257.
Costa Rica Sources
Costa Rican forestry information including common names and distribution.
Peninsula de Osa tree inventory entry.
Area de Conservacion Guanacaste species page.
Burger (2000). Fieldiana: Botany n.s. no. 41, pp. 127-128. Detailed morphological description and keys.
Related Reading
Biography of the Scottish botanist who transferred the species to Jacaranda in 1823.
Biography of the French pharmacist-botanist who first described the species from French Guiana.
Demographic and spatial genetic structure on Barro Colorado Island. Molecular Ecology 15: 3205-3217.